Prepare the transfer plate
Before you begin preparing your reagents, please ensure the Base Fluid has been connected to the instrument overnight. If the Base Fluid was not connected in advance, please open a new vial and incubate it for at least 1 hour at 30°C before use
Take all the reagents out of the freezer. The preparation of the transfer plate takes about 1 hour.
Our Quick Start User Guide can be used as a short guide tool how to prepare the transfer plate
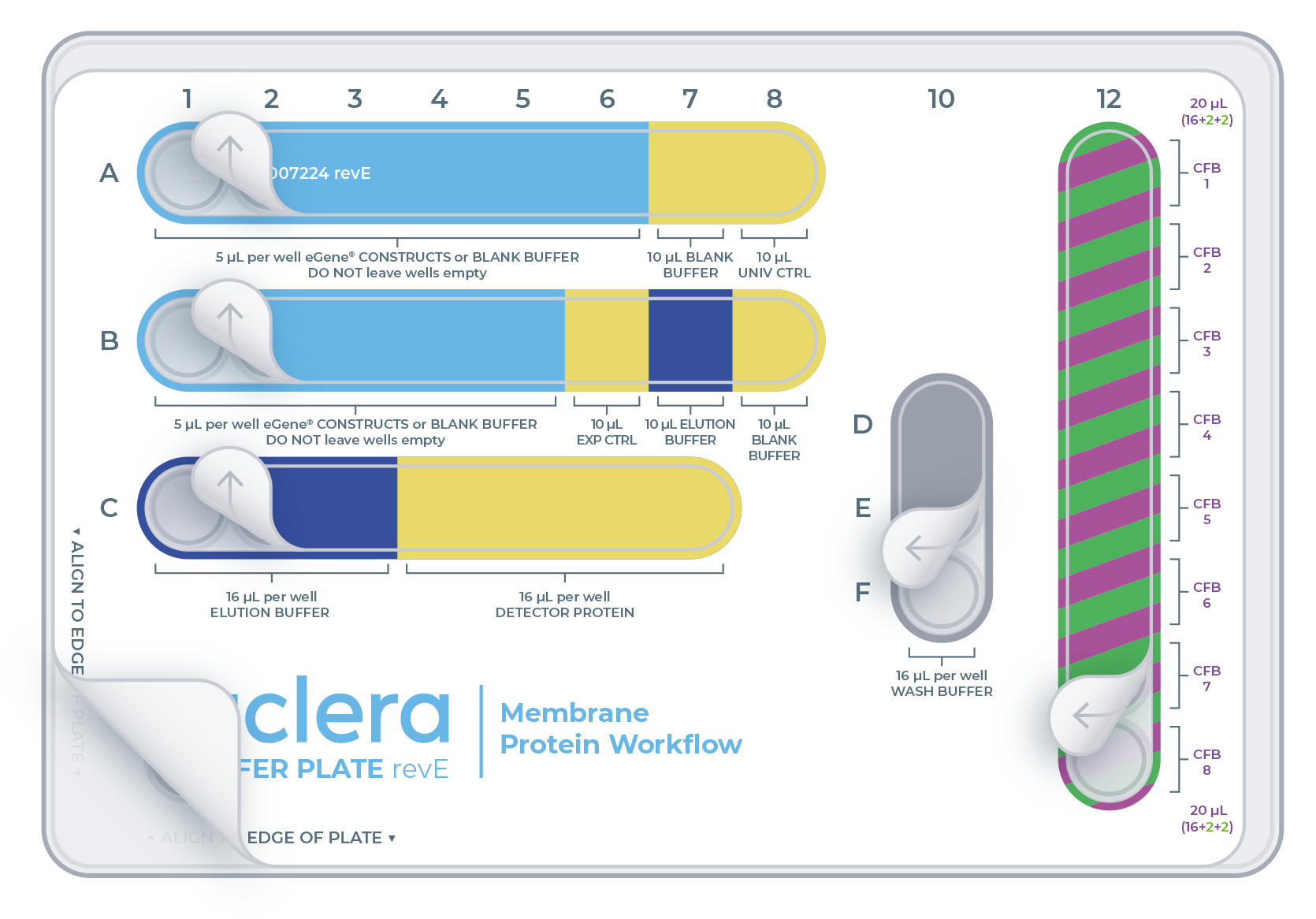
The eProtein Discovery™ reagents need to be prepared and loaded onto a 96 well transfer plate following the layout and volumes in figure and table below.
It is critical to follow this layout exactly because it determines how the reagents are dispensed in the eProtein Discovery™ cartridge.
If an eGene™ construct is missing it must be substituted with 5 µL of Blank Buffer.
Do not substitute a missing eGene™ construct with water as this can have a significant negative impact on the droplet operations on the eProtein Discovery™ cartridge
eGene™ DNA construct
| Reagent | Well | Volume (µL) |
|---|---|---|
| eGene™ construct | A1 - A6 | 5 |
| eGene™ construct | B1 - B5 | 5 |
Reagents from NC3013
| Reagent | Well | Volume (µL) |
|---|---|---|
| Blank Buffer | A7 and B8 | 10 |
| Universal Control (Univ. Ctrl) | A8 | 10 |
| Detector Protein (Det. Prot.) | C4 and C7 | 16 |
| Expression Control | B6 | 10 |
| Wash Buffer | D10, E10 and F10 | 16 |
| Elution Buffer | B7 | 10 |
| Elution Buffer | C1-C3 | 16 |
| Cell-free Core (16 µL) + Additive 1 (2 µL) + Additive 2 (2 µL ) | 12A - 12H | 20 (16+2+2) |
12A-12H
Reagents and volumes to load on the transfer plate1.Take the Strep Beads from the fridge and the Cartridge Kit reagents (box with the purple stripe on the label) from the -80°C freezer.
Place an empty 96-well transfer plate on ice.
Apply the transfer plate sticker provided in the Cartridge Reagent kit and place the 96 well plate on ice.
The transfer plate should be kept on ice until the transfer of reagents to the Cartridge.
Ensure you prepare the Cell-free Blends last.
Be careful not to introduce any ice into the wells
eGene™ constructs (DNA)
Take the vials or the plates with the eGene™ constructs made in advance using the eGene™ Prep Kit out of the freezer and thaw on the benchtop at room temperature. This takes approximately 15 minutes.
The vials or the plates should be centrifuged for a few seconds to ensure all the liquid is at the bottom of the wells.
For every eGene™ construct, load 5 µL into the selected well:
- A1 to A6
- B1 to B5
It is critical to load the eGene™ constructs onto the transfer plate in the exact order that they have been finalized in the experiment planned in the eProtein Discovery™ Cloud Software if using a Cloud Connected System.
eProtein Discovery™ purification reagents
Thaw the Wash Buffer and the Elution Buffer on the benchtop at room temperature. This will take about 20 minutes.
- Load 16 µL of Wash Buffer into wells D10, E10 and F10
- Load 16 µL of Elution Buffer into wells C1, C2, C3
- Load 10 µL of Elution Buffer into well B7
eProtein Discovery™ controls
From the kit kept at -80°C, take the controls out and thaw them on ice.
- Load 10 µL of Blank Buffer into wells A7 and B8.
- Load 10 µL of Universal Control into well A8.
- Load 10 µL of Expression Control into well B6
Strep Purification Beads
Strep Purification Beads are provided in 2x 200 µL aliquots of 5% v/v suspension – To prepare the Strep Beads:
- Take the 2x vials of Strep Beads from the fridge and give them a quick spin for 2 seconds in a microcentrifuge to ensure all material is collected at the bottom of the tubes.
- Resuspend the beads by gently pipetting up and down 10 times with a p200 pipette set on 90 µL.
- Transfer 3x 90µL (2x 90 from one tube and 1x 90 from the second tube) of the resuspended beads into a 3x 1.5 mL tubes. Beads settle quickly - be sure to resuspend between aliquots. Discard the rest only after the experiment starts, in case more volume is required.
- Place the three tubes with Strep Beads on a magnetic particle separator and capture for 1 min.
- Remove all the supernatant with a p200 pipette and discard the liquid.
- Remove the three tubes with Strep Beads from the magnetic particle separator. Resuspend the beads in 100 µL Wash Buffer by slowly pipetting up and down 10 times.
- Repeat steps 4 to 6 twice more for a total of three washes.
- After the third wash, spin down the three tubes and place it back on a magnetic particle separator and capture for 1 min.
- Remove all the supernatant with a p200 pipette and discard the liquid.
- Spin down the tube, place it back on a magnetic particle separator and remove the residual buffer with a p20 pipette.
- With a p20 pipette, resuspend the beads in the three tubes with 10.5 µL of Wash Buffer by gently pipetting up and down 10 times to create three beads solutions of 15 µL at 30% v/v Strep Beads working solution
- Keep the beads in the tube on the bench, not on ice.
The beads should NOT be loaded onto the transfer plate.
Detector Protein
Spin down the tubes for 2 seconds to collect the full volume at the bottom. Load 16 µL of Detector Protein into wells C4, C5, C6 and C7 of the transfer plate as instructed on transfer plate label.
Preparation of the Cell-free Blends
For each expression screening experiment, up to eight distinct 20 µL Cell-free Blends can be prepared by combining 16 µL of Cell-free Core Reagent with 2 µL of a first additive and 2 µL of a second additive. If fewer combinations are used, fill the remaining wells of Column 12 of the transfer plate using 16 µL of Cell-free core + 4 µL of Additive Buffer.
The total volume of blend should always be 20 µL final.
Ensure that the Cell-free blend is thoroughly resuspended by pipetting up and down from near the bottom.
- Thaw Cell-free Core Reagents and Additives on ice
- Once the Cell-free Core reagents and Additives are thawed, vortex each for 2 seconds to ensure they are well mixed.
- Centrifuge for 2 seconds the Cell-free Core reagents and Additives using a microcentrifuge to return any droplets to the bulk aliquot.
- Add 16 µL of Cell-free Core reagent to wells A12-H12.
- Add 2 µL of your first selected additive to wells A12-H12.
- Add 2 µL of your second selected additive to wells A12-H12.
It is critical to load the Cell-free Blends onto the transfer plate in the exact order that they have been finalized in the experiment planned in the eProtein Discovery™ Cloud Software.
Ensure the Cell-free Blend is thoroughly resuspended with the chosen additives by pipetting up and down near the bottom of the tube, making sure that any viscous components are fully mixed.